Microglia-neuron interactions regulate brain development, maturation, homeostasis, and disease genesis. The innate immune complement component C1q, produced and secreted by microglia, has brain function specificity. C1q acts as an initiating molecule for the classical complement pathway and mediates synaptic pruning related to development and disease. Interestingly, in the aging brains of humans and rodents, C1q protein levels are significantly upregulated, while other complement proteins remain at lower levels. This suggests that C1q may have an age-dependent function that is independent of the classical complement pathway.
During aging, C1q accumulates throughout the brain parenchyma, synapses, and GABAergic interneurons. In C1q-deficient mice (C1qKO), synaptic plasticity is enhanced in the hippocampal regions, ameliorating cognitive and memory decline. However, the knockdown of C1q enhances plasticity without resulting in synaptic pruning mediated by the complement pathway. In the cortex of aging animals, C1q is distributed within neuronal synaptic terminals, axons, and dendrites, suggesting that the age-dependent function of C1q may be mediated through neurons.
On June 27, 2024, Beth Stevens’ team at the Center for Neurobiology at Boston Children’s Hospital published an article in Cell titled “Microglial-derived C1q integrates into neuronal ribonucleoprotein complexes and impacts protein homeostasis in the aging brain.” They found that microglia-derived C1q regulates the translation of specific mRNAs in a phase-separated manner by integrating into neuronal ribonucleoprotein (RNP) complexes, impacting protein homeostasis and cognitive function in the aging brain.
Microglia-Secreted C1q Co-localizes with Neuronal RNP Complexes in an Age-Dependent Manner
Scientists isolated synaptosomes from developing (5 days), adult (2-3 months), and aged (1 year) mice and analyzed them by mass spectrometry after immunoprecipitation with a C1q antibody (C1q-IP). They found that C1q protein interactions changed dramatically during the aging process. Specifically, during development, C1q mainly interacted with extracellular functional proteins, whereas during aging, C1q primarily interacted with intracellular active proteins. Further experiments revealed that C1q was enriched in ribosomes and interacted with the ribosomal protein RPL10a.
Immunofluorescence revealed distinct punctate C1q signals within the neuronal cytosol, and these signals were more localized to neurons in an age-dependent manner. Using mice with fluorescently labeled neuronal ribosomes to examine the interaction between complement C1q and RNP complexes within neurons, the team found that C1q co-localized with the neuronal RNP complexes.
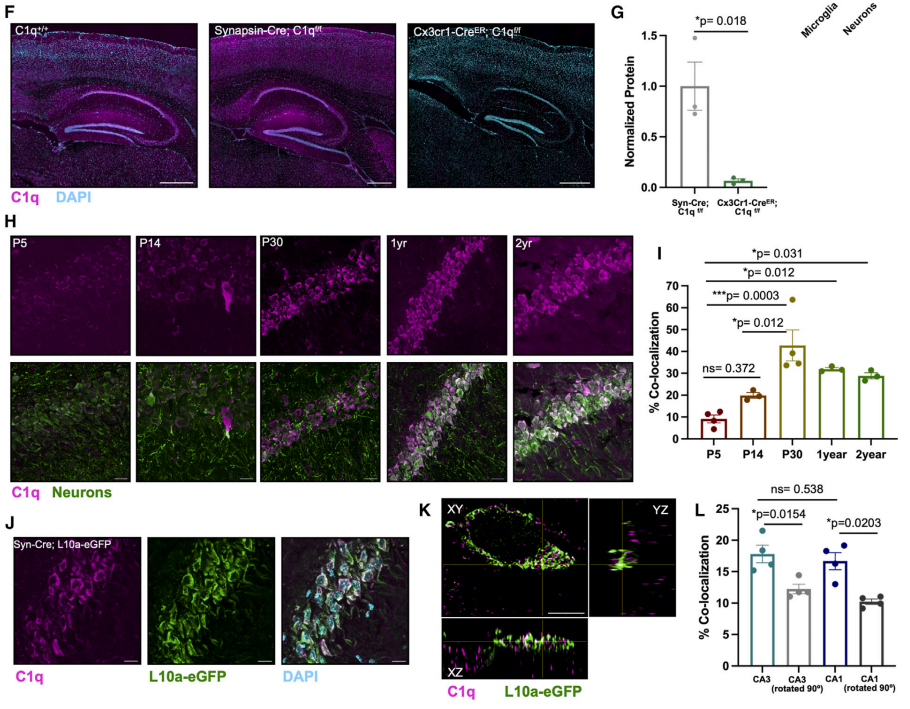
Fig. 1 C1q co-localizes with neuronal RNP complexes in an age-dependent manner.1
C1q-RNP Complexes Regulate mRNA Translation by Liquid-Liquid Phase Separation
Liquid-liquid phase separation (LLPS) occurs when the collagen-like domain of C1q interacts with the RNP complex. LLPS droplets can form when purified C1q protein binds to RNA isolated from human brain tissue, and these droplets disappear after treatment with RNase, suggesting that droplet formation depends on both C1q and RNA. Further experiments revealed that exogenous C1q protein integrates into the neuronal RNP complex in an endocytosis-dependent manner.
Using WT and C1qKO littermates, the team examined the effect of knocking out C1q on neuronal mRNA translation in vivo at different ages. No significant differences in neuronal mRNA translation in vivo were detected during the developmental stage (5 days) and adulthood (2-3 months). However, in old age (1 year), C1qKO mice showed significant changes in mRNA translation compared to their WT littermate.
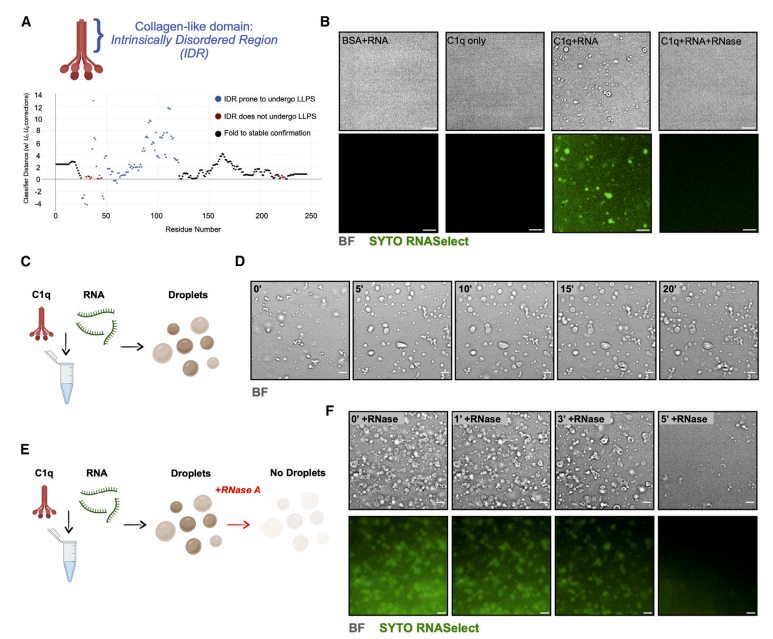
Fig. 2 C1q-RNP complexes regulate mRNA translation by liquid-liquid phase separation.1
Microglia-Derived C1q Affects Protein Translation and Fear Memory Dissolution in an Age-Dependent Manner
Whole-brain proteomic analysis revealed significant differences in protein content between the brain tissues of WT and C1qKO littermates. The brain tissues of WT mice were enriched in Septin complex-associated proteins, which regulate dendritic spine formation, axon growth, synaptic vesicle release, and ion channel localization. In contrast, C1qKO mouse brain tissue was enriched in mitochondrial proteins. An examination of learning and memory capacity in aged WT and C1qKO littermate mice found that microglia-specific knockdown of C1q did not affect the acquisition and extraction of fear memories but significantly inhibited the dissipation of fear memories.
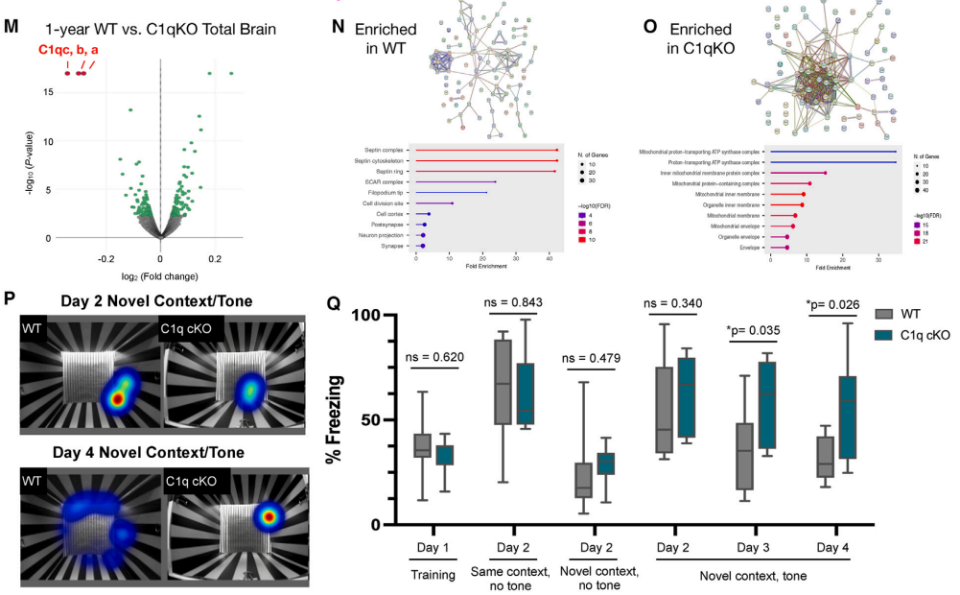
Fig. 3 Microglia-derived C1q affects protein translation and fear memory dissipation in an age-dependent manner.1
This study found that microglia-derived C1q integrates into neuronal RNP complexes in an age-dependent manner, regulating neuronal protein translation and homeostasis, thereby affecting cognitive function in the aging brain.
Reference:
- Scott-Hewitt, Nicole, et al. “Microglial-derived C1q integrates into neuronal ribonucleoprotein complexes and impacts protein homeostasis in the aging brain.” Cell(2024).
