Recently, Elios Therapeutics released positive top-line data to evaluate the clinical trial of TLPLDC (tumor lysate, particle loading, dendritic cell) vaccine in the treatment of melanoma in phase IIb.
1.Research result
This is a prospective, randomized, double-blind, placebo-controlled trial in patients with stage III (advanced) or IV (metastatic) melanoma who have been resected to evaluate the efficacy and safety of the TLPLDC vaccine. The primary endpoint of the study was a two-year disease-free survival (DFS), secondary endpoint, including 36 months of DFS and overall survival (OS), will be compared between the TLPLDC vaccine group and the placebo group. In this study, 144 patients were randomly given TLPLDC or placebo to prevent recurrence of the disease. TLPLDC vaccine or placebo vaccine was vaccinated, and at 0, 1, 2, 6, 12 and 18 months, respectively, within 3 months after the completion of standard nursing (SoC) treatment. The recurrence rate was followed up according to SoC. In view of the high early recurrence rate in high-risk melanoma patients and the time it takes for vaccines to activate the immune system, the main efficacy of intentional therapy (intent-to-treat, ITT) and treatment (per treatment, PT) populations were analyzed.
The results showed that the study reached the main endpoint, and in the PT group, the TLPLDC vaccine significantly reduced the risk of disease recurrence compared with placebo at the 24th month of treatment. The specific data were as follows: in all 144 patients in the study group (ITT), including those who had never or had not been fully vaccinated, the recurrence rate was 66% in the placebo group and 54% in the TLPLDC vaccine group. This means that the risk of recurrence is 18% lower in clinical significance, although not statistically significant, but reduces the relative risk of recurrence. However, PT analysis of all patients who had completed primary immunization with TLPLDC vaccine or placebo vaccine (6 months) showed a recurrence rate of 56% in the placebo group and 29% in the TLPLDC vaccine group, which represents a statistically significant 50% reduction in the relative risk of disease recurrence.
The Independent data Security Monitoring Committee (DSMB) assessed the results of the study and determined that there were no safety issues, and only 1/3 of patients experienced related adverse events, most of which were level 1 or 2 (AE). In addition, a preliminary assessment of 36-month follow-up data for all patients showed that the benefits of the TLPLDC vaccine were not only lasting but still beneficial 24 months later. As a result, DSMB recommends that the study continues in accordance with the agreement until the end of the 36-month DFS and OS milestones, which are expected to be reached in June 2020.
The full results of the study will be presented at an upcoming medical conference. Mark B.Faries, director of surgical oncology and co-director of the melanoma program at the Los Angeles Clinic and Research Institute, said: “as a therapist, significantly reducing the risk of cancer recurrence is an important task for me and my patients. It is an exciting possibility to be able to use patients’ own tumors to create a custom vaccine that allows them to be immune to their own cancer. Data from the IIb phase study showed strong clinical benefits, with a 50% reduction in the risk of recurrence in patients who completed the initial immunization of the vaccine. In addition, the data confirm the significant safety of the vaccine and increase its attractiveness as a potential new treatment for patients at high risk of recurrence. “
Buddy Long, CEO of Elios Therapeutics, said: “High-risk melanoma is an invasive disease that requires safer and more effective treatment. This is the first active phase IIb clinical trial of individualized cancer vaccine therapy for high-risk melanoma patients. We believe that the results of this trial support the promotion of the TLPLDC vaccine clinical project to phase III. “
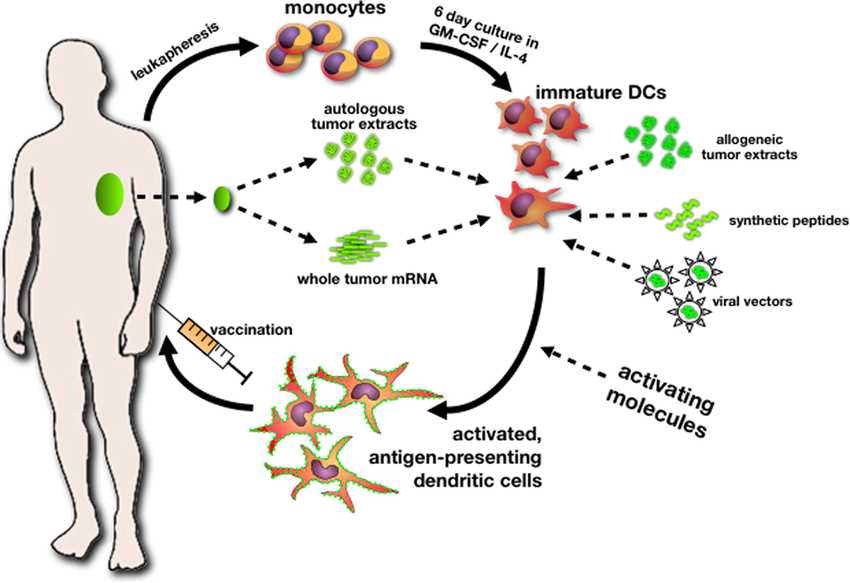
2. Mechanism of TLPLDC vaccine
TLPLDC is an autologous therapeutic cancer vaccine made from patients with their own cancer cells. The aim is to stimulate the immune system to identify tumor cells and fight patient-specific (or unique) cancer: (1) this immunotherapy presents tumor antigens in particles with pathogen-associated molecular characteristics (PAMPs); (2) the presenting pathogen carrying tumor antigen triggered congenital and adaptive dual immune response, (3) secreting high levels of IL-12 and IL-15, to produce high-affinity CD8 cytotoxic T cells.
TLPLDC (tumor lysate, granule loading, dendritic cell) vaccine is unique immunotherapy, both in manufacturing and delivery. The vaccine is personalized, which means it is made from the patient’s tumors and blood. Each patient’s tumor has a unique antigenic feature, and dendritic cells in the blood are the most effective antigen-presenting cells in the body. Once injected with TLPLDC, it transports all of the patient’s tumor antigen library to the immune system, producing a dual innate and adaptive immune response that activates T cells. It also triggers the immune system to identify, and destroy any cells that contain antigens and specific mutations derived from their tumors.
Phase IIb clinical trials of TLPLDC vaccine for advanced melanoma are being conducted at major academic cancer centers in the United States as monotherapy and standard care combined with checkpoint inhibitors.
