A small-scale clinical trial suggests that an improved form of CAR-T cell therapy, known as Descartes-08, holds promise for treating myasthenia gravis, an autoimmune neuromuscular disease. The modified CAR-T therapy used in this trial showed potential for more durable reduction of myasthenia gravis symptoms with good tolerability and no significant adverse reactions. The findings of this study were published in the July 2023 issue of Lancet Neurology, titled “Safety and clinical activity of autologous RNA chimeric antigen receptor T-cell therapy in myasthenia gravis (MG-001): a prospective, multicentre, open-label, non-randomized phase 1b/2a study.”
Dr. Emily Caporello, Director of Small Business Programs at the National Institute of Neurological Disorders and Stroke, stated, “Repurposing breakthrough therapies like CAR-T to treat a neurological disorder demonstrates the versatility of immunotherapy when treatment options are limited or nonexistent.”
Myasthenia gravis is a chronic autoimmune disease most commonly caused by the immune system attacking a protein at the neuromuscular junction. The hallmark of this disease is muscle weakness that worsens with activity and improves with rest. Current treatment focuses on symptom management, primarily targeting muscle weakness.
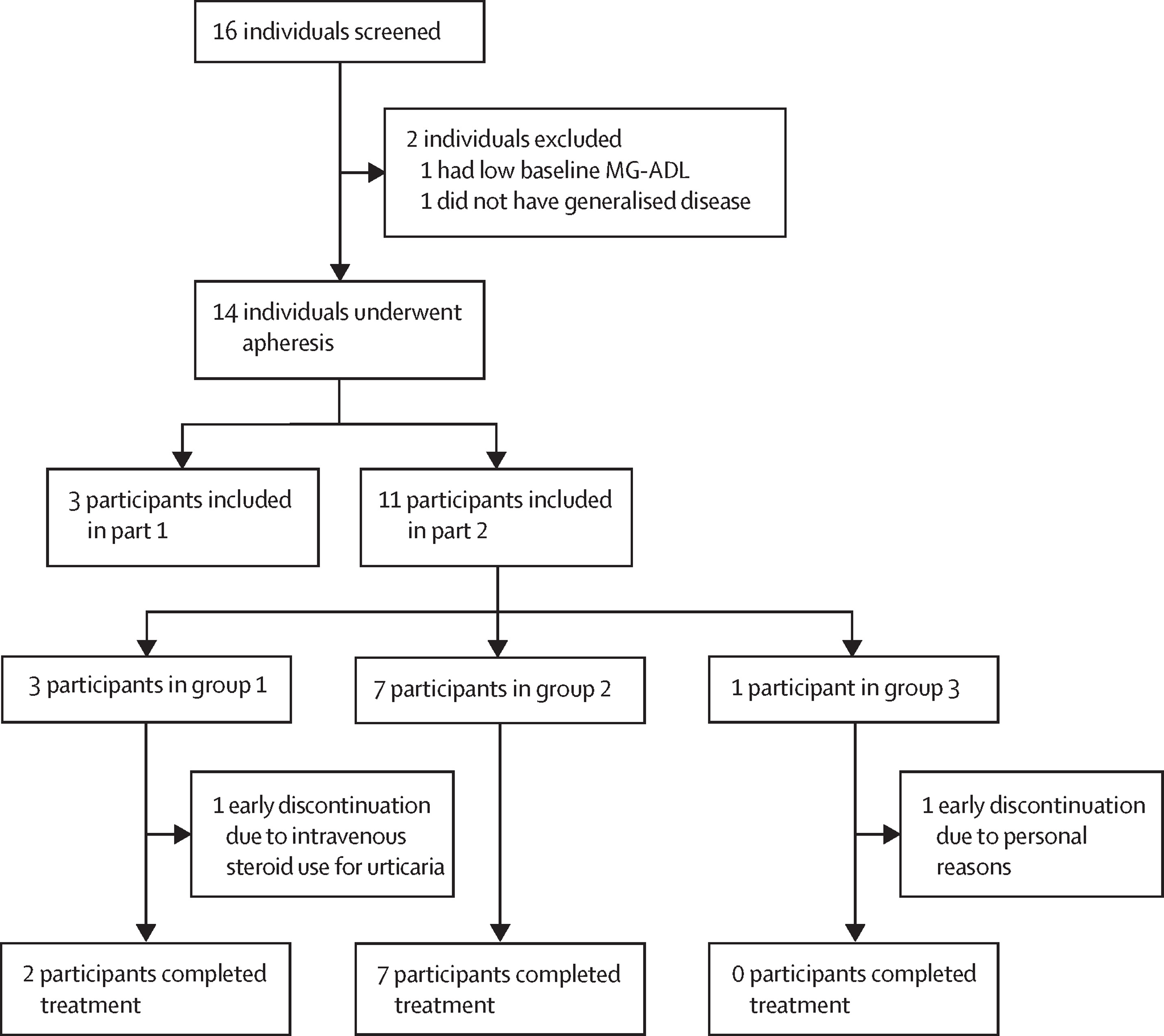
In this new study, 14 patients with generalized myasthenia gravis received varying doses of the modified CAR-T cell therapy called Descartes-08, which targets the cells responsible for producing the antibodies that cause myasthenia gravis. The optimal dosing was determined to be once weekly for six weeks.
Early data on the efficacy of this cellular therapy is promising, but further clinical research is needed to assess its effectiveness. Three patients receiving Descartes-08 treatment showed complete or near-complete elimination of their symptoms, which persisted for six months after treatment. Additionally, two patients no longer required long-term intravenous immunoglobulin therapy, currently used for severe cases of myasthenia gravis.
Dr. Murat V. Kalayoglu, President and CEO of Cartesian Therapeutics, stated, “We are seeing profound and enduring responses to Descartes-08, lasting at least six months post-treatment. We have now initiated a larger randomized, placebo-controlled clinical trial, the first for an engineered allogeneic cell therapy.”
CAR-T cell therapy involves extracting a patient’s T cells—the key players of the immune system that can recognize and destroy invading pathogens—and reprogramming them to target a specific antigen. In the case of blood cancers, the new target is the cancer itself. For myasthenia gravis, the target is the damaged cells producing the destructive antibodies.
Many immunotherapies, including CAR-T cell therapy, carry risks of severe side effects, which are tolerable in advanced cancer cases but prohibited in chronic diseases like myasthenia gravis. Typically, T cells are reprogrammed using DNA, which can persist in cells and lead to amplified effects and serious adverse reactions.
To avoid such side effects, Descartes-08 uses messenger RNA (mRNA) instead of DNA to reprogram the T cells, as mRNA does not replicate during cell division. This results in a shorter treatment regimen administered multiple times, as opposed to the single-dose approach used in CAR-T cell therapies that rely on DNA programming. The primary goal of this clinical trial was to determine the ideal dose of Descartes-08 for effectively reducing myasthenia gravis symptoms while minimizing side effects.
Currently, Descartes-08 cellular therapy is undergoing a larger clinical trial to determine its ability to reduce myasthenia gravis symptoms. Importantly, this trial will also include a placebo group, a crucial control to confirm any observed improvements are due to the cellular therapy and not influenced by unrelated factors.
Reference
1. Granit, Volkan, et al. “Safety and clinical activity of autologous RNA chimeric antigen receptor T-cell therapy in myasthenia gravis (MG-001): a prospective, multicentre, open-label, non-randomised phase 1b/2a study.” The Lancet Neurology 22.7 (2023): 578-590.