Salmonella Strains-based Ames Fluctuation Test
Creative Biolabs offers Salmonella strains-based Ames fluctuation test service in ADME. This test is a bacterial analysis that has a high predictive value for rodent carcinogenicity tests.
Ames Test
The Ames test is a widely used method for assessing the mutagenic potential of chemical compounds. It was developed by Dr. Bruce Ames and colleagues in the 1970s and is based on the observation that certain strains of Salmonella typhimurium can revert to a histidine-positive phenotype when exposed to mutagens. The test is commonly used as a first step in evaluating the genotoxic risk of chemicals and has been accepted by international organizations.
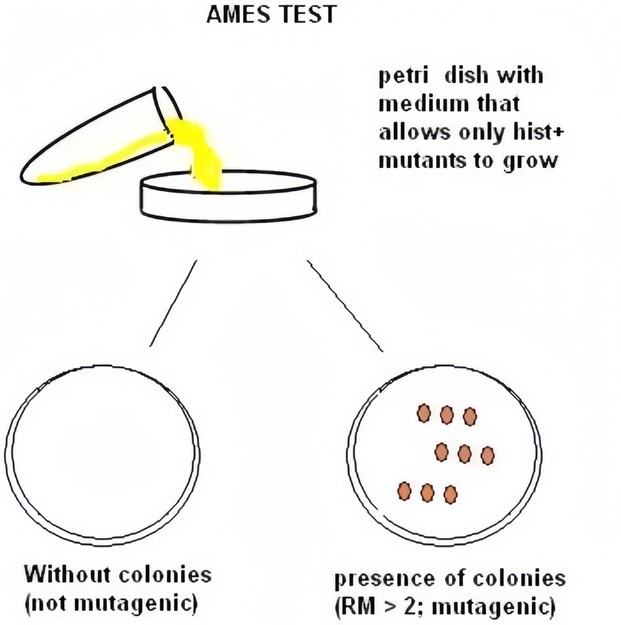 Fig.1 A brief summary of the Ames test.1
Fig.1 A brief summary of the Ames test.1
The Ames test involves exposing the bacterial strains to the test compounds and then observing whether they induce mutations in the bacteria. The test can detect different types of mutations, such as frameshift mutations and base pair substitutions, and is sensitive to chemicals that are carcinogenic to rodents and potentially to humans. While the test is highly sensitive, it may also produce false positives and false negatives, so it is often used in conjunction with other genotoxicity tests for a more comprehensive assessment.
This widely accepted method has several advantages as below:
-
High Sensitivity: The test is highly sensitive and can detect chemicals that are carcinogenic to rodents and potentially to humans. This sensitivity makes it effective for screening suspected compounds and identifying mutagens.
-
Pharmaceutical Development: The pharmaceutical industry commonly uses the Ames test during the development of new therapeutic drugs. Positive results in the test can lead to the termination of drug development, indicating its importance in identifying genotoxic hazards early in the drug development process.
-
Metabolism Studies: The Ames test can be utilized to study xenobiotic metabolism and DNA repair systems. By incorporating liver homogenates from rodents, the test can evaluate the role of enzymes like CYP in metabolizing pre-mutagens, providing insights into metabolic pathways.
-
Epidemiological Studies: The test can be employed in epidemiological studies to assess human populations exposed to occupational mutagens, environmental contaminants, and other hazardous substances. Urinary mutagen detection using the Ames test has been valuable in evaluating chronic exposures to mutagens.
Salmonella Strains-based Ames Fluctuation Test
Creative Biolabs offers Salmonella strains-based Ames fluctuation test service with four Salmonella strains (TA98; TA100; TA1535; TA1537) with and without metabolic activation by rat liver S9. It is recommended to incubate the samples at 37°C.
Tab.1 Specific information on four Salmonella strains.
|
Strain
|
Mutation
|
Target DNA
|
R-factor
|
|
TA98
|
hisD3052
|
CGCGCGCG
|
+R
|
|
TA100
|
hisG46
|
GGG
|
+R
|
|
TA1535
|
hisG46
|
GGG
|
-R
|
|
TA1537
|
hisC3076
|
TAA
|
+R
|
Ames fluctuation test is a valuable tool for assessing the mutagenic potential of chemical compounds, typically involves the following steps:
Notes:
-
Plate the bacteria on agar plates that lack histidine, as only the revertant bacteria that have undergone mutations and regained the ability to synthesize histidine will be able to grow on these plates.
-
Depending on the results of the Ames test, further testing may be conducted to confirm the mutagenic potential of the compounds and assess their genotoxic risks comprehensively.
If you are interested in the salmonella strains-based Ames fluctuation test, please contact us, and we will provide you with high-quality service as soon as possible. And you might be interested in these related services: High-Throughput ADME, Analysis of Protein Binding, and Analysis of Drug-Drug Interactions.
Reference
-
Dos Santos, J. L., et al. "Mutagenicity of new lead compounds to treat sickle cell disease symptoms in a Salmonella/microsome assay." International journal of molecular sciences 11.2 (2010): 779–788.
For Research Use Only | Not For Clinical Use


 Fig.1 A brief summary of the Ames test.1
Fig.1 A brief summary of the Ames test.1

 Download our brochure
Download our brochure