Age Related Macular Degeneration (AMD)
Retinal gene therapy holds great promise for the treatment of inherited and noninherited blinding diseases such as retinitis pigmentosa and age-related macular degeneration (AMD). It has revolutionized AMD treatment by drastically reducing administration frequencies. With access to advanced instruments, fit-for-purpose laboratories, and professional knowledge, Creative Biolabs has become a leading, fully integrated biopharmaceutical company focused on next-generation gene therapy. Our expertise has been widely proven in therapeutic vectors' design, optimization & construction, delivery vehicles development, potency tests, safety and toxicology analysis, solutions of specific gene therapy development for diseases. Our mission is to work closely with clients to achieve long-term mutually beneficial relationships.
Background of AMD
AMD is a degenerative disease of the retina that mainly affects the macula and is a leading cause of irreversible blindness worldwide. It can broadly be categorized into two forms, non-neovascular and neovascular (nAMD). The former is characterized by the development of drusen and retinal pigment epithelial (RPE) changes in early disease (usually called dry AMD), and with loss of RPE and associated severe vision loss in advanced disease. While the latter is mainly characterized by choroid neovascularization (CNV), which causes macular exudation leading to fibrosis (usually called wet AMD), RPE atrophy, and finally permanent central vision loss.
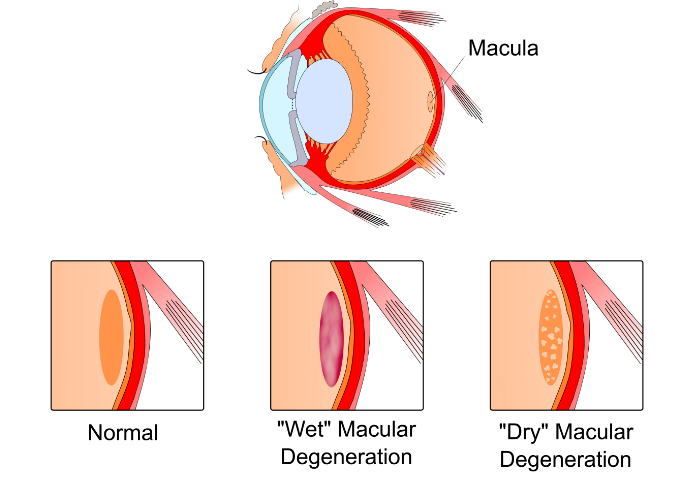 Figure 1. Age related macular degeneration (AMD).
Figure 1. Age related macular degeneration (AMD).
Current Status of AMD Treatment
There are no available curative options for advanced dry AMD. Studies demonstrate that the complement system may play a role in the development of dry AMD. CD59, an inhibitor of terminal complement complex [membrane attack complex (MAC)], is regarded as a therapeutic target for dry AMD. For wet AMD, current therapies mainly aim to prevent the growth of the abnormal retinal blood vessels. This growth inhibition is achieved by monthly intravitreal injections of molecules that block the activity of vascular endothelial growth factor (VEGF). However, monthly bolus therapy with anti-VEGF agents causes cyclical effects on vision and center point thickness (CPT) of the macula, as well as enormous and growing cost burden. Therefore, an alternative therapy that could keep continuous anti-VEGF suppression is clearly needed.
Gene Therapy for AMD
Gene therapy can revolutionize AMD treatment by drastically reducing administration frequencies. By inserting an anti-VEGF complementary DNA construct encoded in a viral vector into photoreceptors and retinal pigment epithelial cells, it can upregulate the VEGF inhibitor production and block the activity of VEGF. The most widely used vectors for ocular gene delivery are adeno-associated virus (AAV) because they elicit minimal immune responses and mediated long-term transgene expression in a variety of retinal cell types. Rakoczy, et al. had conducted an adeno-associated viral (AAV) vector that delivered the gene encoding soluble Fms-related tyrosine kinase-1 (sFLT-1), a naturally occurring inhibitor of VEGF to achieve long-term VEGF blockade. In addition, gene therapy is being assessed to chronically express anti-angiogenic proteins such as pigment epithelium-derived factor (PEDF), as well as RAR related orphan receptor A (RORA) gene, angiostatin and endostatin. Currently, there are nine rAAV-based gene therapy products for AMD listed in development, of which 4 in clinical trials and 5 in preclinical development.
 Figure 2. Lifecycle of recombinant AAV.sFLT-1. (MacLaren, 2015)
Figure 2. Lifecycle of recombinant AAV.sFLT-1. (MacLaren, 2015)
What We Can Do at Creative Biolabs?
With years of development, Creative Biolabs provides flexible solutions about gene therapy through our offer to select stand-alone services or executing projects in an integrated manner. We have developed a comprehensive library of gene delivery vectors, including various viral and no-viral vehicles, which can transfer precise amounts of foreign gene products to each target cell, sequentially allowing expression without causing cytotoxicity. In addition, based on these vehicles, we can perform design & optimization & construction, potency tests, safety and toxicology analysis, etc. to meet basic research and preclinical applications. Our well-established GTOnco™ platform supports both routine and custom-designed assays to meet your immuno-oncology (I-O) drug development program. Aided by this platform, we can especially reverse the inhibition of adaptive immunity, switch on or expand adaptive immunity and so on.
Features
- Advanced strategies for rAAV vector design and construction
- Optimized rAAV vector engineering techniques
- Highly customized services to meet specific demands
- Best after-sale service
AAV-based gene therapy has achieved great development in the past decades in ocular. Creative Biolabs is dedicated to providing high-quality rAAV vector-based AMD services, utilizing our advanced molecular strategies and experienced engineering techniques to get a perfect vector for gene therapy. Inquired for details are appreciated, please feel free to contact us to get more information.
Reference
- MacLaren, R.E. (2015). Gene therapy for age-related macular degeneration. Lancet. 386(10011):2369-70.
