In the realm of molecular interactions, two terms that frequently arise are avidity and affinity. These terms are pivotal in understanding how molecules, such as antibodies, antigens, and ligands, interact with each other. While they might appear similar at first glance, avidity and affinity have distinct meanings and play unique roles in the world of biochemistry and molecular biology. This article will delve into the differences between avidity and affinity, shedding light on their significance in various biological contexts.
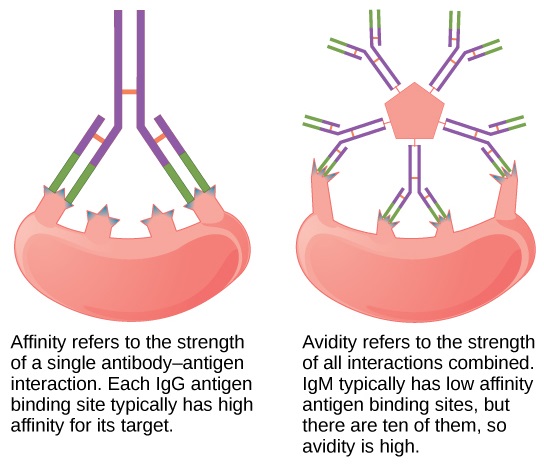 Fig.1 Difference Between Affinity and Avidity.
Fig.1 Difference Between Affinity and Avidity.
Avidity is a term used to describe the overall strength or stability of a molecular interaction, typically between multiple binding sites or multiple interactions between molecules. It's a measure of the cumulative strength of multiple bonds or interactions occurring simultaneously. Avidity takes into account factors like the number of binding sites on one or both interacting molecules and the strength of individual bonds.
One of the most common examples of avidity in biology is the binding of antibodies to antigens. Antibodies are Y-shaped proteins with two identical antigen-binding sites, allowing them to bind to antigens with multiple epitopes. The simultaneous binding of both antigen-binding sites to multiple epitopes on the same antigen increases the overall avidity of the interaction. This multivalent binding enhances the stability of the antibody-antigen complex.
Avidity can be influenced by several factors:
Valency: The number of binding sites or interacting domains on the molecules involved. A molecule with multiple binding sites can achieve higher avidity through multivalent interactions.
Bond Strength: The strength of individual bonds formed between the interacting molecules. Stronger bonds contribute more to the overall avidity.
Concentration: The concentration of molecules in the solution can influence the likelihood of multiple interactions occurring simultaneously.
Affinity, on the other hand, refers to the strength of a single binding interaction between two molecules, typically a receptor and its ligand. It quantifies how tightly a receptor and ligand bind to each other at a specific binding site. Affinity is a fundamental concept in molecular biology and is crucial for understanding various biological processes, including enzyme-substrate interactions, receptor-ligand interactions, and more.
Affinity can be influenced by the following factors:
Molecular Shape: The complementarity in the shape and chemical properties of the binding sites on the interacting molecules. A better fit leads to higher affinity.
Electrostatic Interactions: Attractive or repulsive forces between charged or polar molecules can affect affinity.
Hydrophobic Interactions: The interaction between nonpolar regions of molecules can also influence binding affinity.
Temperature and pH: These environmental factors can affect the strength of affinity between molecules.
Avidity: A measure of the overall strength or stability of a molecular interaction, taking into account multiple binding sites or interactions.
Affinity: The strength of a single binding interaction between two molecules at a specific binding site.
Avidity involves multiple interactions occurring simultaneously, often due to multivalent binding.
Affinity focuses on a single, specific binding interaction.
Avidity depends on valency, bond strength, and concentration of molecules.
Affinity is affected by molecular shape, electrostatic interactions, hydrophobic interactions, and environmental factors.
Avidity is commonly seen in interactions involving multivalent molecules, such as antibodies binding to antigens.
Affinity is crucial in various biological processes, including enzyme-substrate interactions, receptor-ligand interactions, and drug-receptor interactions. (Learn more about our Protein-Protein Interaction (PPI) Assay Services)
Avidity is not easily quantified and is often assessed experimentally.
Affinity is quantified using equilibrium dissociation constants (Kd).
Understanding the distinctions between avidity and affinity is crucial in several biological processes, and these concepts often intersect, influencing the outcomes of various molecular interactions:
Vaccine Development: Avidity is a critical factor in vaccine design. Vaccines aim to elicit a strong and long-lasting immune response. By engineering antigens that can induce antibodies with high avidity for a pathogen, vaccines become more effective at providing immunity.
Antibody Maturation: During the course of an infection, the avidity of antibodies can change. Initially, antibodies may have low avidity, but over time, as the immune system matures, they can develop higher avidity, providing better protection against reinfection.
Tissue Development: Avidity-driven cell adhesion is instrumental in embryonic development and tissue formation. Cells use various adhesion molecules with multivalent interactions to ensure proper tissue organization and function.
Cancer Metastasis: In cancer biology, changes in cell adhesion avidity can have significant implications. Alterations in the avidity of cancer cells for the extracellular matrix or other cells can promote metastasis, enabling cancer cells to invade distant tissues.
Metabolic Regulation: Enzyme-substrate affinity plays a pivotal role in metabolic regulation. Enzymes with high substrate affinity can fine-tune metabolic pathways, ensuring efficient utilization of nutrients and energy production.
Biotechnology: In biotechnological applications, enzymes with specific affinities are employed for various purposes, such as DNA polymerases in PCR or restriction enzymes in genetic engineering.
Therapeutic Efficacy: Affinity-driven drug design is central to the development of targeted therapies. High-affinity drugs can selectively bind to their intended target proteins, modulating their function with precision while minimizing off-target effects.
Resistance Mechanisms: Understanding how drug affinity can change over time is crucial in combating drug resistance. Pathogens or cancer cells can evolve to reduce the affinity of their target proteins for drugs, rendering therapies less effective.
Synaptic Transmission: In the nervous system, the affinity of neurotransmitters for their receptors governs synaptic transmission. Precise control of affinity ensures the accurate transmission of signals between neurons, underpinning cognitive processes.
Neurodegenerative Diseases: Alterations in the avidity and affinity of molecules involved in neurodegenerative diseases, such as Alzheimer's and Parkinson's, can contribute to the pathogenesis of these conditions. Understanding these changes is critical for developing therapeutic interventions.
Signal Transduction: Affinity is a key factor in cell signaling pathways. Ligand-receptor interactions with high affinity trigger specific cellular responses, leading to processes like cell proliferation, differentiation, and apoptosis.
Hormone Regulation: Hormones, such as insulin and thyroid hormones, exhibit high affinity for their respective receptors. Dysregulation in hormone-receptor affinity can lead to metabolic disorders.
In the realm of molecular biology and biochemistry, avidity and affinity are two distinct but interconnected concepts that govern how molecules interact. Avidity represents the cumulative strength of multiple interactions, while affinity quantifies the strength of a single interaction. Both concepts are indispensable in understanding a wide range of biological processes, from immune responses to enzymatic reactions and drug development. Recognizing their unique roles helps researchers and scientists better comprehend the intricacies of molecular interactions and harness this knowledge for various applications in science and medicine.
All listed services and products are For Research Use Only. Do Not use in any diagnostic or therapeutic applications.
| USA:
Europe: Germany: |
|
|
Call us at: USA: UK: Germany: |
|
|
Fax:
|
|
| Email: info@creative-biolabs.com |
